CUPRAC antioxydant capacity
Reference : KF01005
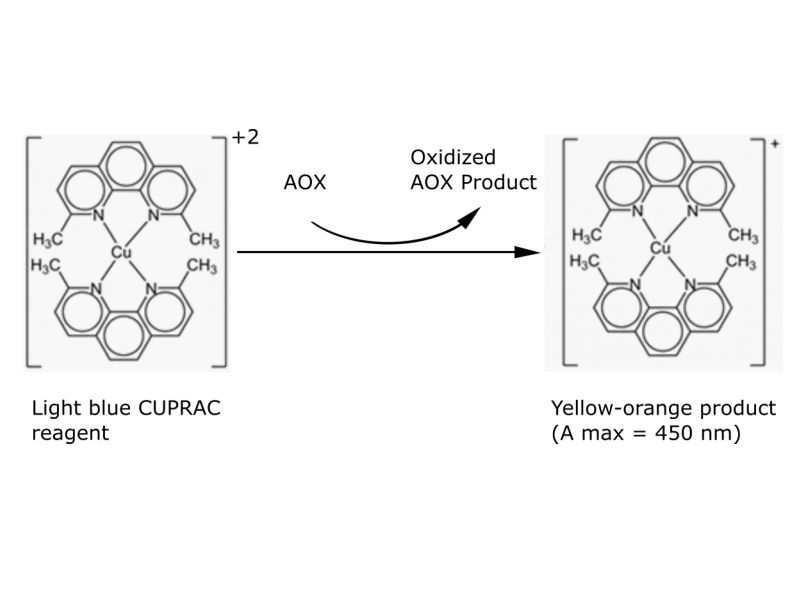
The main reagent, the copper (II)-neocuproin (2,9-dimethyl-1,10-phenanthroline), can oxidize antioxidants generating a coloured product measurable by colorimetric methods. The chelation with neocuproin enables a fastened reaction by elevating the redox potential of the reagent.
Recent correlations have been saw within many illnesses and ROS (reactive species of oxygen, as superoxide); antioxidants on the other hand have been proved to inhibit this ROS. So the measure of these antioxidants seems an interesting new approach to study not only illnesses, also many other physiological states.
Assay Principle
CUPRAC reagent implies an oxidation of an antioxidant with a leading tiol group, like for example gluthation (GSH). In this process, the reagent reduces itself forming a chelate complex of cupper (I) – neocuproin, wich provides color measurable at 450nm in a spectrophotometer. This reaction takes place on physiological pH (or neutral pH, near pH 7).
The documents available in this section are provided for information only and are subject to change. Please contact us for the latest updates.
If you have just received a kit or product without technical instructions in paper format, this means that the instructions have been dematerialized.
In this case, please download it and refer only to the version you can download from the product page of the manufacturer's website, or by scanning the QR code present on the product.
Regarding any certificates of analysis, safety data sheets or spreadsheets, please contact us to obtain the latest versions.
If you have any questions, our team is at your disposal.
Specifications
| Reference | Packaging | |
|---|---|---|
| KF01005-100 | 100 tests (96 puits/wells format) | |
| KF01005-200 | 200 tests (96 puits/wells format) | |
| KF01005-400 | 400 tests (96 puits/wells format) |